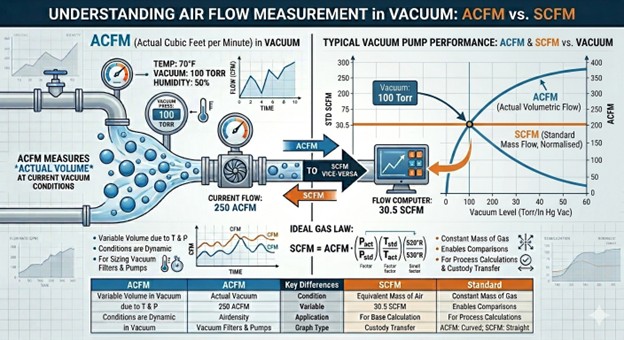
The answer is, they’re both right! But they are different.
SCFM is essentially a molar flow rate disguised as a volumetric one. While a regular volumetric flow rate (ACFM or “Actual” CFM) changes if the gas gets hot or compressed, SCFM is fixed to a specific set of “Standard” conditions {Pstd} and {Tstd). Because those conditions are constant, the Ideal Gas Law proves that SCFM is directly proportional to the molar flow rate ṅ.

Comparison: SCFM vs. ACFM
| Term | Meaning | Is it proportional to ṅ (moles)? |
| ACFM | Actual Cubic Feet per Minute | No. It changes with P and T. |
| SCFM | Standard Cubic Feet per Minute | Yes. It represents a fixed mass/molar flow. |
Summary of Proportionality
For all “non-standard” conditions (ACFM):
- V is proportional to ṅ only if P and T are held constant.
- If P or T change, V is no longer a reliable way to count molecules.
For SCFM:
- V is always proportional to ṅ because it mathematically “undoes” the effects of actual pressure and temperature to tell you how much “stuff” (moles) is actually there.
- 1 SCFM = ṅ = 0.002635 lb-mol/min or 0.1518 lb-mol/hr (molar flow rate)
- 1lb-mol = 2.73×1026 molecules (Avogadro’s number)